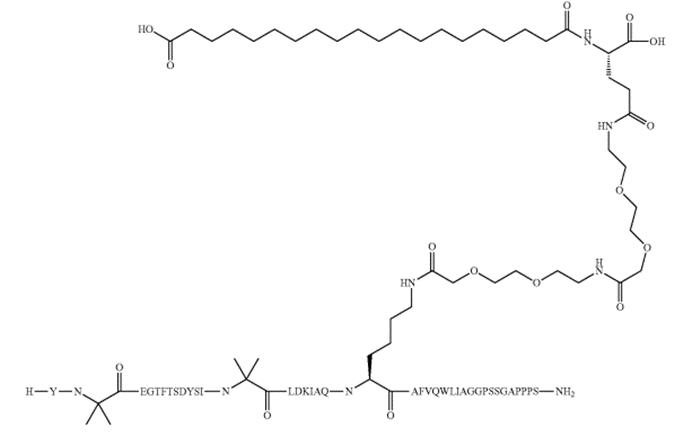
Tirzepatide development is a new medication for the treatment of diabetes and is considered an alternative to Trulicity. It has shown good efficacy in the treatment of type 2 diabetes and has a lower risk of side effects.
Tirzepatide weight loss, improves insulin resistance and cardiovascular risk.Tirzepatide is a long-acting medication that is convenient because it only needs to be injected once a week. This article will cover some of the basics of Tirzepatide.
1. tirzepatide compound Indications
Tirzepatide is primarily used to treat patients with type 2 diabetes, especially those who have had other treatments but have not been able to achieve ideal glycemic control. It can be used as monotherapy or in combination with other oral antidiabetic drugs or insulin. In addition, Tirzepatide is particularly useful for patients with overweight or obesity, as it not only helps to lower blood sugar levels, but also reduces body weight. It suppresses appetite, prolongs satiety, and promotes fat burning, helping to reduce weight and waistline. Studies have shown that Tirz can have a positive impact on the cardiovascular system while controlling blood sugar. It reduces the risk of cardiovascular events and improves lipids, blood pressure and vascular function.
2. Tirzepatide dosage
The dosage of Tirzepatide is very simple, requiring only one subcutaneous injection per week. The choice of dosage should be adjusted according to the patient’s specific situation and the doctor’s advice. In general, a starting dose of 3 mg is recommended, followed by gradual increases to the maximum dose every 4 weeks. The maximum dose of tilpeptide is 15 mg, but it should be used in accordance with the instructions of a physician and individualized to the patient’s needs.
3. tirzepatide side effects
Tirzepatide has shown significant efficacy in clinical trials. It can help patients achieve better glycemic control, reduce insulin resistance, lose weight, and improve cardiovascular risk factors. Tirzepatide has a relatively low side effect profile compared to other similar medications, but may still cause some common adverse reactions.
1. Gastrointestinal Reactions: Common adverse reactions to Tirzepatide include nausea, vomiting, diarrhea, and dyspepsia. Most of these reactions are mild and gradually decrease with treatment.2. Hypoglycemia: Hypoglycemia sometimes occurs when using Tirzepatide. Hypoglycemia is often manifested by symptoms such as dizziness, sweating, palpitations, increased appetite, and difficulty concentrating. If hypoglycemia occurs, it should be treated immediately with appropriate amounts of carbohydrates or as recommended by your doctor.3. Other Adverse Reactions: A small number of people may experience pain, redness, swelling, or skin reactions at the injection site. Rare adverse reactions may include pancreatitis, kidney injury, and cardiovascular events. Report any unusual symptoms or discomfort to your doctor.
Overall, Tirz is an effective, convenient and relatively safe medication for the treatment of type 2 diabetes. It not only helps patients control their blood sugar levels, but also reduces weight and improves cardiovascular risk factors. Before using Tirzepatide, you should consult your doctor for advice and ensure that you know the correct usage and dosage. Its efficacy can only be maximized and potential risks reduced if it is used appropriately under the guidance of a doctor.
Pronunciation /tɜːrˈzɛpətaɪd/
tur-ZEP-ə-tyde
Trade names Mounjaro, Zepbound
Other names LY3298176, GIP/GLP-1 RA
Routes of administration Subcutaneous
Drug class Antidiabetic, GLP-1 receptor agonist
ATC code A10BX16 (WHO) QA10BX16 (WHO)
Bioavailability 80%
Protein binding Albumin
Metabolism Proteolytic cleavage, β-oxidation of fatty diacid section and amide hydrolysis
Elimination half-life 5 days
Excretion Urine and faeces
More Introduction:https://en.wikipedia.org/wiki/Tirzepatide