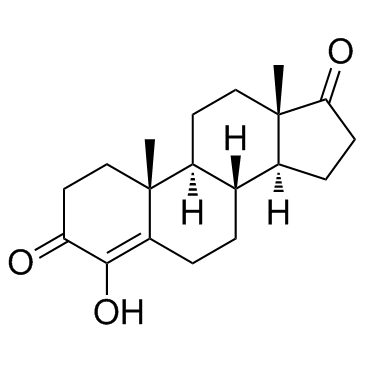
Formestane, formerly sold under the brand name Lentaron among others, is a steroidal, selective aromatase inhibitor which is used in the treatment of estrogen receptor-positive breast cancer in postmenopausal women. The drug is not active orally, and was available only as an intramuscular depot injection. Formestane powder was not approved by the United States FDA and the injectable form that was used in Europe in the past has been withdrawn from the market. It is an analogue of androstenedione.
use
Formestane is often used to suppress the production of estrogens from anabolic steroids or prohormones. It also acts as a prohormone to 4-hydroxytestosterone, an active steroid which displays weak androgenic activity in addition to acting as a weak aromatase inhibitor.
Anti-cancer drugs It also known as lentaron,is an anti-cancer drug, it is primarily used for the treatment of postmenopausal women with advanced breast cancer, it is also effective in prostate cancer.
It is a androstenedione derivative, and it belongs to an aromatase inhibitor with aminoglutethimide, it is a hormone antineoplastic agent. In physiological conditions, it may competitively inhibit the synthesis of the enzyme leading to estrogen biosynthesis decrease in tissues, then it plays its role in cancer. When the tumor tissue growth relies on the presence of estrogen, in order to inhibit tumor growth, the elimination of tumor estrogen-mediated growth stimulation is necessary. This product is more selective than aminoglutethimide while its activity is 100 to 1000 times of aminoglutethimide, and it does not inhibit the synthesis of adrenal hormones,without having to recharge cortisone, etc . The in vitro inhibition of aromatase enzyme of this product is 60 times stronger than aminoglutethimide.
Cooperation
While it is used alone, the drug can not significantly reduce the pre-menopausal estrogen levels in the blood of women,when it is combined with goserelin (gonadotropin-releasing hormone agonist), its inhibitory effect of estrogen in premenopausal women is greater than goserelin used alone. Formestane has no cross-resistance with other aromatase inhibitors , it has no side effects of aminoglutethimide. After oral administration,it is rapidly absorbed by gastrointestinal, its peak plasma concentration time is 1 to 1.5 hours. But the peak concentration of individual is of great difference; after intramuscular injection,it can be accumulated at the injection site and be slowly absorbed. It performs Biphasic elimination process, the initial elimination half-life is 2 to 4 days, the terminal elimination half-life is 5 to 10 days. It is mainly metabolized in the liver after oral administration, in the form of glycosides acid metabolites excreted in the urine.
History
Buy Formestane is a second-generation steroidal aromatase inhibitor, and the first one to reach clinical use during the early 1990s.
Uses An antitumor drug. An aromatase inhibitor.
Uses antineoplastic, aromatase inhibitor
Definition ChEBI: A 17-oxo steroid that is androst-4-ene-3,17-dione in which the hydrogen at position 4 is replaced by a hydroxy group. It was the first selective, type I steroidal aromatase inhibitor, suppressing oestrogen production from anabolic steroids or proho mones. It was formerly used in the treatment of oestrogen-receptor positive breast cancer in post-meopausal women. As it has poor oral bioavailability, it had to be administered by (fortnightly) intramuscular injection. It fell out of use with the subseque t development of cheaper, orally active aromatase inhibitors. It is listed by the World Anti-Doping Agency as a substance prohibited from use by athletes.
Formestane Chemical Properties
Melting point 199-202°C
alpha D20 +181° (c = 7.7 in chloroform)
Boiling point 383.44°C (rough estimate)
density 1.1189 (rough estimate)
refractive index 1.4200 (estimate)
storage temp. 2-8°C
solubility Chloroform (Slightly), Methanol (Slightly)
form solid
pka 9.31±0.60(Predicted)
color White to Off-White
InChI InChI=1S/C19H26O3/c1-18-10-8-15(20)17(22)14(18)4-3-11-12-5-6-16(21)19(12,2)9-7-13(11)18/h11-13,22H,3-10H2,1-2H3/t11-,12-,13-,18+,19-/m0/s1
InChIKey OSVMTWJCGUFAOD-KZQROQTASA-N
SMILES C1(=O)C(O)=C2[C@](C)(CC1)[C@]1([H])[C@]([H])([C@@]3([H])[C@@](CC1)(C)C(=O)CC3)CC2
CAS DataBase Reference 566-48-3(CAS DataBase Reference)
More Introduction:https://en.wikipedia.org/wiki/Formestane